Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Yomogida, Takumi; Esaka, Fumitaka; Magara, Masaaki
Analytical Methods, 9(44), p.6261 - 6266, 2017/11
Times Cited Count:9 Percentile:55.65(Chemistry, Analytical)A combination of micro-sampling, micro-Raman spectroscopy (MRS), and secondary ion mass spectrometry (SIMS) was applied to the characterization of individual uranium particles. Reference particles with U O
O (NBL CRM U010) and UO
(NBL CRM U010) and UO were identified by scanning electron microscopy combined with energy dispersive X-ray detection (SEM-EDX) and transferred onto grassy carbon substrates by micro-sampling. The crystalline phases of the reference particles with diameters ranging from 1
were identified by scanning electron microscopy combined with energy dispersive X-ray detection (SEM-EDX) and transferred onto grassy carbon substrates by micro-sampling. The crystalline phases of the reference particles with diameters ranging from 1  m to 5
m to 5  m were determined non-destructively by using MRS thanks to the optimization of laser power at the measurement. Isotope ratios were also determined with SIMS after the MRS analysis and were consistent with values in the literature. These results indicate that chemical forms and isotope ratios of individual uranium particles as small as 1
m were determined non-destructively by using MRS thanks to the optimization of laser power at the measurement. Isotope ratios were also determined with SIMS after the MRS analysis and were consistent with values in the literature. These results indicate that chemical forms and isotope ratios of individual uranium particles as small as 1  m can be analyzed efficiently by using the proposed method.
m can be analyzed efficiently by using the proposed method.
Yomogida, Takumi; Esaka, Fumitaka; Magara, Masaaki
no journal, ,
Chemical forms of particles in environmental samples give important information on the history of particles. In particular, we can detect the nuclear activities such as refining, conversion and enrichment process by determination of the chemical form of uranium particles. In this study, we developed a micro-Raman spectroscopy technique combined with particle detection by scanning electron microscope (SEM) and micro manipulation to identify chemical forms of standard uranium particles. The characteristic Raman bands which derive from U O
O structures were observed in the Raman spectra of a standard uranium particle with a diameter of 2
structures were observed in the Raman spectra of a standard uranium particle with a diameter of 2  m.
m.
Yomogida, Takumi; Esaka, Fumitaka; Magara, Masaaki
no journal, ,
In this study, we developed a micro-Raman spectroscopy technique combined with alpha track-detection to identify chemical forms of uranium particles. U O
O and UO
and UO particles were used as samples. Films containing uranium-bearing particles were exposed to alpha track detectors (TNF-1) for a few months. The location of uranium particles in the films were identified by observing alpha tracks which caused by spontaneous fission of uranium. The characteristic Raman bands which derive from U
particles were used as samples. Films containing uranium-bearing particles were exposed to alpha track detectors (TNF-1) for a few months. The location of uranium particles in the films were identified by observing alpha tracks which caused by spontaneous fission of uranium. The characteristic Raman bands which derive from U O
O and UO
and UO structures were observed in the Raman spectra of uranium particles.
structures were observed in the Raman spectra of uranium particles.
Yomogida, Takumi; Esaka, Fumitaka; Magara, Masaaki
no journal, ,
Analysis of individual uranium particles in environmental samples from nuclear facilities is useful for detecting nuclear activity. Uranium isotope ratios in the samples are measured to detect nuclear activities related to production of nuclear weapons. We can detect the nuclear activities such as refining, conversion and enrichment process by determination of the chemical form of uranium particles. In this study, we developed a secondary ion mass spectrometry combined with micro-Raman spectroscopy for isotopic and chemical analysis of individual uranium particles. The developed methods have been applied to the analysis of reference uranium materials. As a result, the isotope ratios and the chemical form of individual uranium particle can be analyzed with the proposed method.
Yomogida, Takumi; Esaka, Fumitaka; Magara, Masaaki
no journal, ,
Recently, we are applying micro-Raman spectroscopy to the identification of the chemical forms of individual uranium particles for nuclear safeguards. In this study, a micro-Raman spectroscopy technique combined with particle detection by scanning electron microscope and micro manipulation was developed to identify chemical forms of uranium particles. The characteristic Raman bands at 445 and 1160 cm assigned to UO
assigned to UO structures were observed in the Raman spectrum of a uranium particle with a diameter of 1
structures were observed in the Raman spectrum of a uranium particle with a diameter of 1  m. The particle shape was preserved after the Raman measurement, which indicated the particle was not burned by the laser beam. In conclusion, the chemical forms of individual uranium particles with micro-meter size can be analyzed with the proposed method without damaging the particles.
m. The particle shape was preserved after the Raman measurement, which indicated the particle was not burned by the laser beam. In conclusion, the chemical forms of individual uranium particles with micro-meter size can be analyzed with the proposed method without damaging the particles.
Yomogida, Takumi; Esaka, Fumitaka; Magara, Masaaki
no journal, ,
Chemical state analysis of individual uranium particles in environmental samples is useful tool for deducing the nuclear activity at nuclear facilities. However, coexisting elements in uranium baring particles would interfere with chemical state analysis by micro-Raman spectroscopy. In this study, X-ray detection and micro-Raman spectroscopy were applied in combination to reduce an influence of coexisting elements during the micro-Raman measurement. The method has been applied to the analysis of reference uranium particles and environmental samples. As a result, the chemical form of individual uranium particles can be analyzed thanks to the optimization of the analyzed spot at a micro-Raman measurement. The characteristic Raman band at 445 cm assigned to UO
assigned to UO structures were observed from the uranium-baring particle in an environmental sample.
structures were observed from the uranium-baring particle in an environmental sample.
 -containing iron phosphate glass
-containing iron phosphate glassFujisawa, Masaharu*; Takebe, Hiromichi*; Kobayashi, Hidekazu; Amamoto, Ippei
no journal, ,
High level radioactive waste contains a relatively large amount of MoO . Mo is known to be a difficult element to incorporate in nuclear waste glass such as borosilicate glasses. On the other hand, phosphate glasses is able to contain MoO
. Mo is known to be a difficult element to incorporate in nuclear waste glass such as borosilicate glasses. On the other hand, phosphate glasses is able to contain MoO over a wide and continuous compositional range. In this study, the effect of MoO
over a wide and continuous compositional range. In this study, the effect of MoO addition on properties of iron phosphate glass is studied. The glass transition temperature was evaluated. The immersion tests were performed according to MCC-2 test method due to evaluate the water durability. The additional effects of MoO
addition on properties of iron phosphate glass is studied. The glass transition temperature was evaluated. The immersion tests were performed according to MCC-2 test method due to evaluate the water durability. The additional effects of MoO are discussed in terms of glass structure.
are discussed in terms of glass structure.
Yomogida, Takumi; Esaka, Fumitaka; Miyamoto, Yutaka
no journal, ,
Identification of the chemical forms of individual uranium particles in environmental samples is important tool for deducing the nuclear activity at nuclear facilities. However, there are few reports on the chemical state analysis of sub-micron sized uranium particles. In this study, micro-sampling and micro-Raman spectroscopy were applied in combination to analyze uranium particles with sub-micrometer size. As a result, the chemical form of individual uranium particles with sub-micrometer size can be analyzed non-destructively thanks to the optimization of the laser power at a micro-Raman measurement.
Yomogida, Takumi; Esaka, Fumitaka; Takahashi, Yoshio*; Kitatsuji, Yoshihiro; Miyamoto, Yutaka
no journal, ,
Recently, we are applying micro-Raman spectroscopy (MRS) to the identification of the chemical states of uranium particles in environmental samples. Raman scattering intensity and spatial resolution increase with shortening laser wavelength. However, fluorescence from samples would be interferences in Raman analysis. It is important for Raman analysis to select the suitable laser wavelength. In this study, Raman spectra of uranium oxide particles were measured with two lasers: 532 nm laser and 785 nm laser. UO , U
, U O
O , and UO
, and UO n(H
n(H O) particles were used as samples. No fluorescence was observed in the Raman spectra of the uranium oxide particles. The characteristic Raman bands which derive from UO
O) particles were used as samples. No fluorescence was observed in the Raman spectra of the uranium oxide particles. The characteristic Raman bands which derive from UO , U
, U O
O , and UO
, and UO n(H
n(H O) were observed. As a result, 532 nm laser is suitable for Raman analysis of the uranium oxide particles in terms of sensitivity and spatial resolution.
O) were observed. As a result, 532 nm laser is suitable for Raman analysis of the uranium oxide particles in terms of sensitivity and spatial resolution.
 with
with  -Raman spectroscopy and X-ray absorption spectroscopy
-Raman spectroscopy and X-ray absorption spectroscopyYomogida, Takumi; Ouchi, Kazuki; Matsumura, Daiju; Tsuji, Takuya; Kobayashi, Toru; Kitatsuji, Yoshihiro
no journal, ,
A combination of micro-Raman spectroscopy (MRS), and X-ray absorption spectroscopy was applied to the characterization of uranium dioxide (UO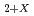 ). A fresh UO
). A fresh UO immediately after preparation, an aged UO
immediately after preparation, an aged UO after 30 years storage were employed. In X-ray absorption near edge structure (XANES) spectra, the absorption edge of the aged UO
after 30 years storage were employed. In X-ray absorption near edge structure (XANES) spectra, the absorption edge of the aged UO sample was shifted to higher energy. This result indicates that the aged UO
sample was shifted to higher energy. This result indicates that the aged UO was slightly oxidized. Characteristic Raman bands at 447, and 1150 cm
was slightly oxidized. Characteristic Raman bands at 447, and 1150 cm which derived from UO
which derived from UO structure were observed in both the flesh and the aged UO
structure were observed in both the flesh and the aged UO . The ratio of a Raman peak at 1150 cm
. The ratio of a Raman peak at 1150 cm to at 447 cm
to at 447 cm of the fresh UO
of the fresh UO is higher than that of the aged UO
is higher than that of the aged UO . These results are showing the applicability of the proposed methods to analyzing the oxidation of UO
. These results are showing the applicability of the proposed methods to analyzing the oxidation of UO .
.
 -Zr-SS system
-Zr-SS systemKusaka, Ryoji; Kumagai, Yuta; Watanabe, Masayuki; Akiyama, Daisuke*; Kirishima, Akira*; Sato, Nobuaki*; Sasaki, Takayuki*
no journal, ,
Fuel debris is considered to alter over time due to the contact with water in the reactors of 1F nuclear power station. The chemical state of the fuel debris is related to its stability, and can be important information for removal, storage, and processing-and-disposal of the fuel debris. Recently, we have conducted immersion tests of uranium-zirconium-stainless steel simulated debris in hydrogen peroxide solution to investigate the reaction with hydrogen peroxide generated by radiolysis of water. As a result, the elution behavior of uranium suggested that the surface of the simulated debris was altered by the immersion. In this study, the chemical state of the debris surface altered by the immersion was identified using micro-Raman spectroscopy. In addition, the existence distribution of the alteration phase near the surface was investigated.
Yomogida, Takumi; Kitatsuji, Yoshihiro; Takahashi, Yoshio*; Miyamoto, Yutaka
no journal, ,
In the decommissioning of the FDNPP, there is a concern about the release of fine particles containing uranium (U) into the environment. Since the solubility of U in water varies greatly depending on its chemical species, it is inevitable to understand the chemical species of U fine particles. Uranium dioxide (UO ) is oxidized to a non-stoichiometric compound (UO
) is oxidized to a non-stoichiometric compound (UO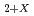 ) by air oxidation. In this study, we developed a method to discriminate the chemical species of the bulk of microparticles by transmission X-ray absorption spectroscopy and of the surface of microparticles by micro-Raman spectroscopy. Aged UO
) by air oxidation. In this study, we developed a method to discriminate the chemical species of the bulk of microparticles by transmission X-ray absorption spectroscopy and of the surface of microparticles by micro-Raman spectroscopy. Aged UO particles were used as a model compound. By analyzing the X-ray absorption spectra, it was found that UO
particles were used as a model compound. By analyzing the X-ray absorption spectra, it was found that UO and U
and U O
O were present in about 90% and 10% of the fine particles, respectively. In the Raman spectra, the Raman peaks derived from the structure of UO
were present in about 90% and 10% of the fine particles, respectively. In the Raman spectra, the Raman peaks derived from the structure of UO nH
nH O were observed, suggesting that the main chemical species are different between the surface and the bulk of the particles.
O were observed, suggesting that the main chemical species are different between the surface and the bulk of the particles.